The Science of Climate Change
1. Climate System
The purpose of this section is to explain the climate system. An understanding of how the climate system works to maintain the Earth's energy balance is necessary to understand how the changes we see in our environment are forcing warming and other related changes that are collectively known as climate change.
Climate System
What controls Earth’s climate?
The image below is an illustration of the interdependent aspects of the Earth's climate system relative to the Earth's energy balance.
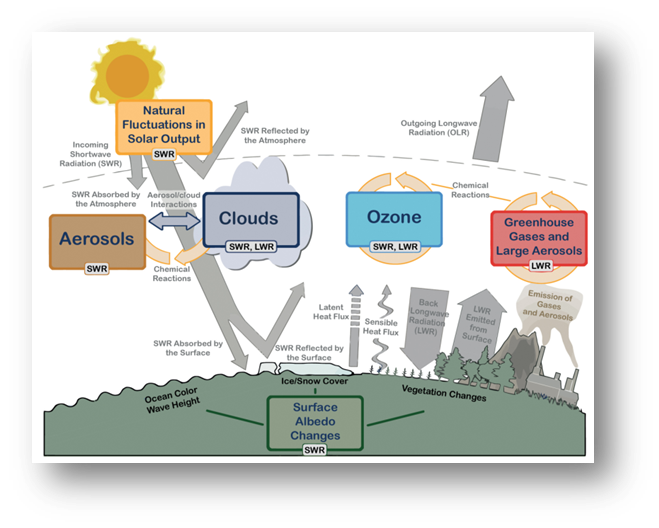
Source: Cubasch, U., D. Wuebbles, D. Chen, M.C. Facchini, D. Frame, N. Mahowald and J.-G. Winther, 2013: Introduction. In: Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change [Stocker, T.F., D. Qin, G.-K. Plattner, M. Tignor, S.K. Allen, J. Boschung, A. Nauels, Y. Xia, V. Bex and P.M. Midgley (eds.)]. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA, pp. 119–158, doi:10.1017/CBO9781107415324.007.
The video below provides an overview of the Earth's climate system.
The climate of the Earth is controlled by the Earth’s energy balance, which is the movement of energy into and out of the Earth system.
Energy flows into the Earth from the Sun and flows out when it is radiated into space.
The Earth’s energy balance is determined by the amount of sunlight that shines on the Earth (insolation) and the characteristics of the Earth’s surface and atmosphere that act to reflect, circulate and re-radiate this energy.
This video describes how NASA monitors the Earth's energy flows: https://gpm.nasa.gov/education/videos/real-world-monitoring-earths-energy-budget-ceres
Insolation
Insolation is the measure of the amount of solar radiation (energy) falling on a surface and it is a very important factor in determining the climate of the Earth.

Source: http://www.srh.noaa.gov/jetstream/tropics/tropics_intro.htm
On Earth we notice the effects of varying insolation on our climate. Sunlight falls most directly on the equator, and only obliquely (at an angle) at the poles. This means that sunlight is more concentrated at the equator. As shown in the above figure, the same amount of energy covers twice as much area when it strikes at an angle of 30 degrees compared to when it strikes a surface directly. The energy striking the Earth indirectly has less ability to warm the Earth because there is less energy per unit of surface area than when the sunlight strikes the surface of the Earth more directly.
This difference explains why the equator has a hot climate and the poles have a cold climate. Differences in insolation also explain the existence of seasons.
For many years now, satellites have been monitoring these energy flows using spectrometers that measure the intensity of energy at different wavelengths flowing to the Earth and from the Earth.
The image below shows the insolation at the top of the atmosphere averaged for the month of March.
Approximately 49% of the insolation is absorbed by the Earth’s surface:
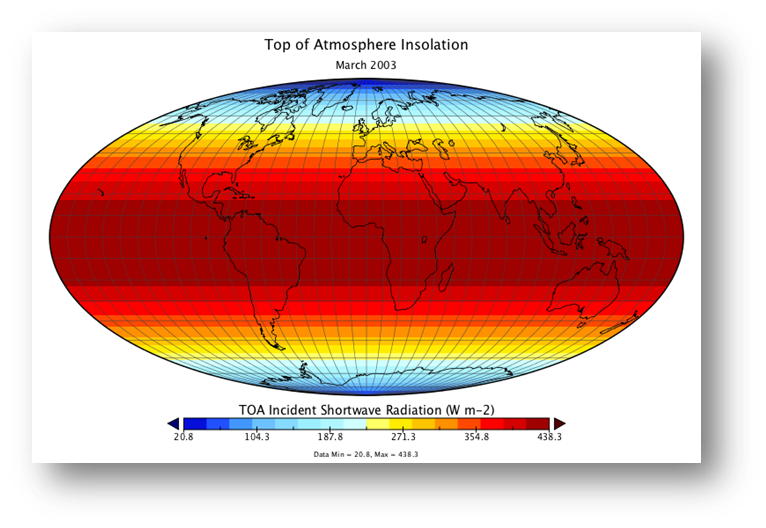
Source:https://courses.ems.psu.edu/earth103/node/1007
The Earth’s orbit around the sun is not perfectly circular so sometimes the earth is closer or further from the Sun. This also changes the amount of insolation, as the closer the Earth is to the Sun the more concentrated the solar radiation. Earth’s orbit is also not fixed, it changes over time. These changes are referred to as Milankovitch Cycles (changing the total insolation and the timing of the insolation).
These orbital variations have made a big difference in conditions on the Earth during the period in which humans have inhabited it.
Energy Flows in the Climate System
The view of the climate system depicted in the figure below is one of stability —energy flows in and out, in perfect balance, so the temperature of the earth should stay the same.
But if we can learn anything from studying Earth’s history, we learn that change is the rule and stability the exception. When change occurs, it almost always brings feedback mechanisms into play — they can accentuate and dampen change and they are incredibly important to our climate system.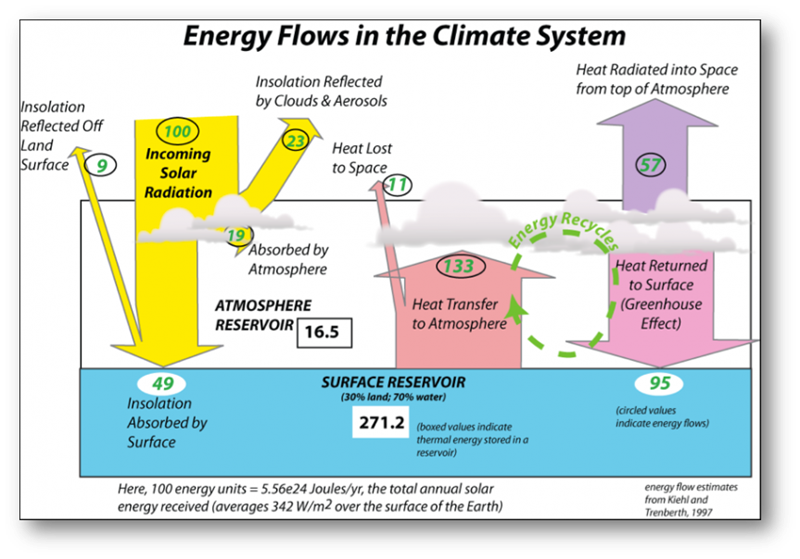
Source: https://www.e-education.psu.edu/earth103/node/1007
Albedo
One way to change how quickly energy exits the Earth system is to change the reflectivity of the surface.
Compare the difference in darker surfaces to the brightness of snow‐covered ice.
The dark surface is going to absorb the sun’s rays (and heat the Earth’s surface) while the snow is reflecting the sunlight back into space.
Albedo is a measure of how reflective a surface is (higher the albedo = the more reflective).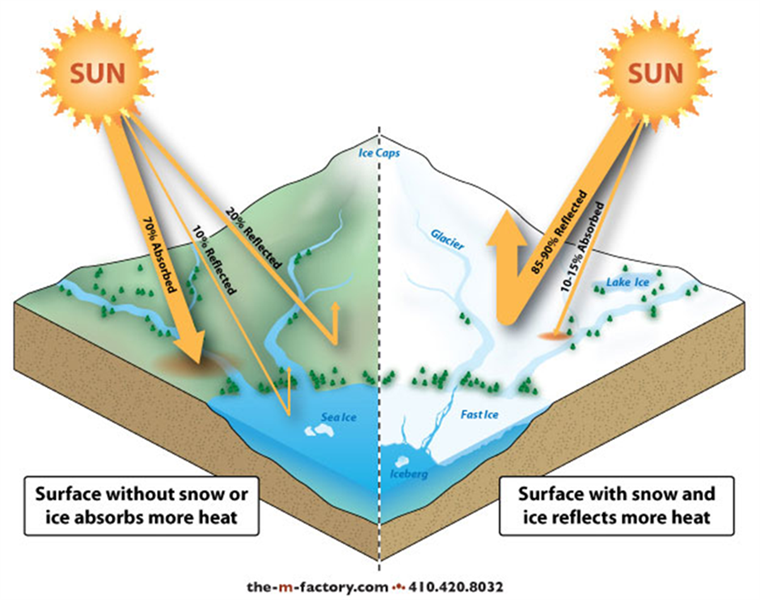 Source: https://oceanbites.org/sea-ice-and-albedo-should-we-be-worried/
Source: https://oceanbites.org/sea-ice-and-albedo-should-we-be-worried/
The video below provides an explanation of the albedo effect.
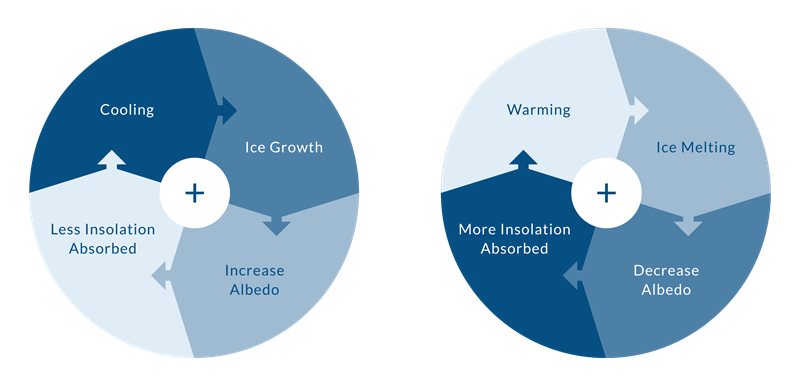
Changes in albedo can create a positive feedback that reinforces a change in the climate. A positive feedback is a process which amplifies the effect of an initial change.
POSITIVE FEEDBACK MECHANISM
If the climate cools (initial change), snow covers more of the surface. Because snow has a higher albedo than bare ground, this initial cooling increased the amount of sunlight that is reflected back into space, cooling the Earth further.
If such a loop continued for long enough, this process could result in the entire Earth being covered in ice.
Since this chain of events furthers the initial change that triggered the whole thing, it is called a positive feedback (but note that the change may not be good from our perspective). Positive feedback mechanisms tend to lead to runaway change — some small initial change is thus accentuated into a major change.
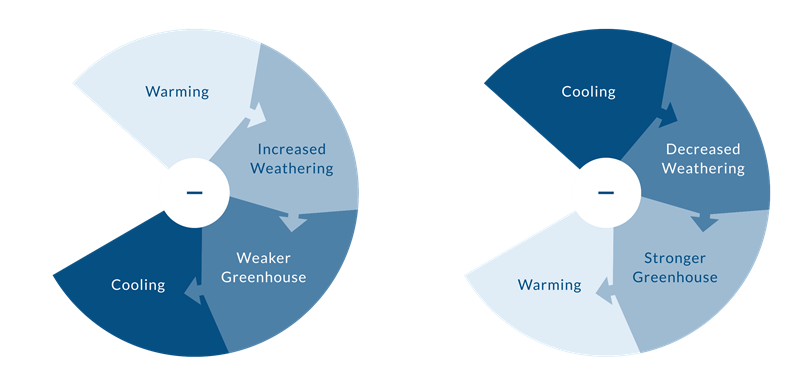
In the Earth system, positive and negative feedbacks are essential components that play an important role in maintaining a more or less stable state. A negative feedback mechanism stabilizes a system, preventing an extreme state.
NEGATIVE FEEDBACK MECHANISM
Weathering Feedback
Rocks exposed at earth’s surface interact with water and the atmosphere undergoing a set of chemical and physical changes called weathering.
The weathering process involves a chemical reaction. When carbon dioxide dissolves in water, carbonic acid is formed. Acid rain contains carbonic acid and when it falls on rocks sensitive to carbonic acid, another chemical reaction is initiated, dissolving minerals and breaking down the structure of the rocks. In this way, the atmospheric carbon is transported by rivers into oceans where it is trapped for thousands of years before it is returned to the atmosphere or stored in marine sediments or corals.
This process of weathering is thus a sink for atmospheric carbon dioxide, which is an important greenhouse gas. If you remove carbon dioxide from the atmosphere, you weaken the greenhouse effect and this leads to cooling of the Earth.
Like many chemical reactions, this chemical weathering occurs more rapidly in hotter climates, which are associated with higher levels of carbon dioxide. So consider a scenario in which some warming occurs; this will encourage faster weathering, which will consume carbon dioxide, which will lead to cooling. In this case, the initial change triggered a set of processes that countered the initial change — this is called a negative feedback (even though it may have beneficial results) because it works in opposition to the change that triggered it.
Climate System
Climate System
- Video released by Yale Climate Connections This Is Not Cool http://www.yaleclimateconnections.org/2013/11/new-ipcc-report-featured-in-this-months-this-is-not-cool-video/
- IPCC Fifth Assessment Report is available here https://www.ipcc.ch/
- This IPCC Report provides information about observed changes in the climate system http://www.climatechange2013.org/images/report/WG1AR5_SPM_FINAL.pdf
- A University of British Columbia online course http://www.youtube.com/user/climateliteracy Of particular interest related to this section are the following:
- Module 2 Introduction to the Climate System https://www.youtube.com/playlist?list=PLAHtWWrZTgn-i_U-ft6cjsTj2faylPSJO
- Module 3 Earth's Energy Balance https://www.youtube.com/playlist?list=PLAHtWWrZTgn8boLKB4-a1PV5y_u_b0MJU
- 100 Views of Climate Change, Colorado State University http://changingclimates.colostate.edu/
2. Greenhouse Effect
2.1 Atmosphere & Greenhouse Gases
The natural greenhouse effect is necessary for the temperature of earth to support current life systems. The enhanced greenhouse effect caused by a greater buildup of carbon dioxide and methane (and other greenhouse gases) leads to less radiated heat leaving the atmosphere, resulting in warmer global temperatures. Scientists are confident that human burning of fossil fuels is a major contributor to the enhanced greenhouse effect.
World population is increasing, industrialization is expanding globally, income levels are rising in newly industrialized countries spurring greater levels of consumption. These realities are contributing to even more rapid increases in greenhouse gases.
Atmosphere & Greenhouse Gases
The Earth’s energy balance is regulated by the atmosphere. The atmosphere is made up of gases. These gases regulate energy transmission through the atmosphere. This is where greenhouse gases come into the picture.
[As an educator, be aware that some students believe that our atmosphere is only the clouds and above the clouds. It is essential for them to know that the atmosphere starts at the surface of the Earth and extends more than 300 miles outward from the surface of the Earth.]
The illustrations below show the Earth's atmosphere visually and then diagramatically.

Image Source: NASA http://earthobservatory.nasa.gov/IOTD/view.php?id=7373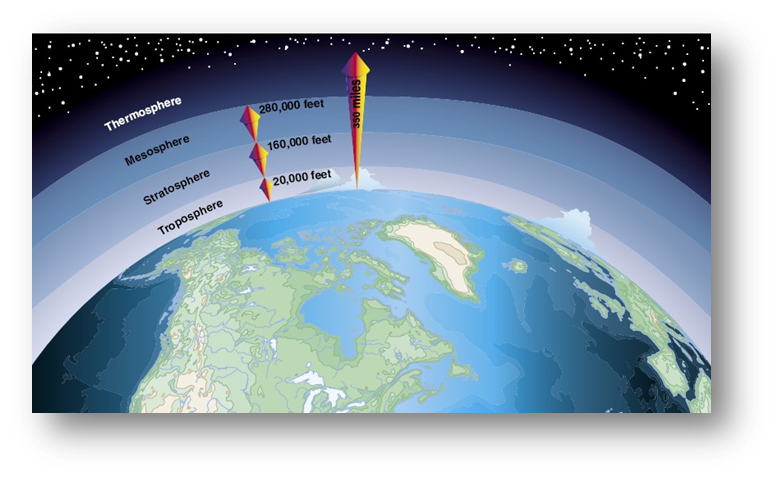
Source: http://www.groundinstructor.com/mod/page/view.php?id=970
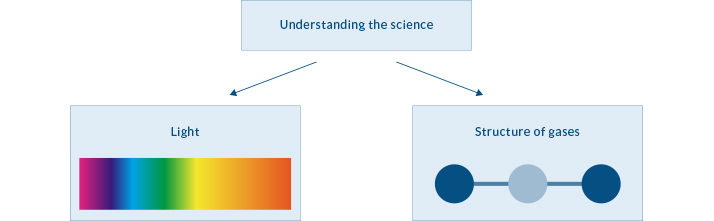
Understanding the Science of the Greenhouse Effect
In order to understand the basic science of the greenhouse effect, two concepts must be explained: the science of light and the structure of the gases in our atmosphere.
Understanding the Science of Light
Let’s begin with light. We’ll use “light” here to refer to all electromagnetic radiation (EMR) which includes many kinds of light such as ultraviolet light, infrared light, X-rays, microwaves and many more. The illustration above shows the visible light spectrum.
Light can be understood by three basic characteristics: wavelength, frequency and energy.
Under In the Classroom below there is a classroom activity that provides an easy way for students to remember these three basic characteristics.
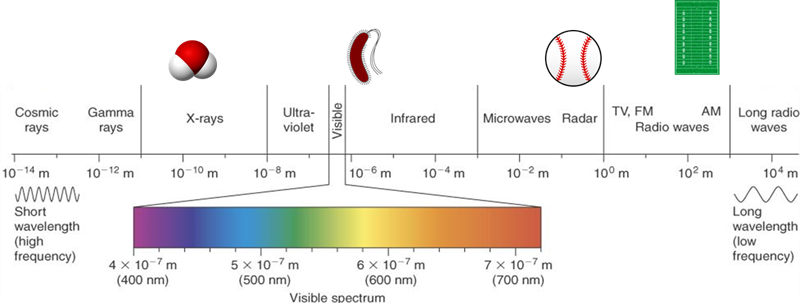
The whole range of EMR from the very short, very energetic cosmic rays to the very long, low energy radio waves acts the same way: Each wavelength has a frequency and energy associated with it.
Visible light spectrum is a small portion of the total type of electromagnetic radiation (EMR). To the left on this illustration there are shorter (invisible) ultraviolet wavelengths with higher frequency and more energy. The longer infrared wavelengths to the right of visible spectrum have lower frequency with less energy.
Light (or EMR) has an incredible variation in wavelength. Very small wavelength and very energetic light like X-rays are very high energy. We know they are high in energy because X-rays can go right through us to make the images of our bones we call “X-rays”! On the other hand, radio and TV waves are huge – often meters long, and of very low energy.
The energy of light (EMR) is fundamentally related to how the Earth's atmosphere traps heat, or the greenhouse effect. Under In the Classroom below there is a classroom activity in the form of a POGIL, which stands for Process Oriented Guided Inquiry Learning. This POGIL provides students the opportunity to examine how the energy in sunlight interacts with the atmosphere to trap heat.
Atmosphere & Greenhouse Gases
- Demonstrating the wavelength, frequency and energy of light (pdf)
- Wavelength Activity - Video Demonstration
- ACTIVITY (POGIL): The Earth's Greenhouse (pdf)
You can learn more about POGIL at: https://pogil.org/about
What is POGIL? from The POGIL Project on Vimeo.
Atmosphere & Greenhouse Gases
- This link to a website called EXPLAINTHATSTUFF! provides an overview of the natural greenhouse effect https://www.explainthatstuff.com/globalwarmingforkids.html
- The Department of Geosciences at Georgia State University has published a set of labs that may be helpful. Of particular interest are the following:
- Lab 2 Stratospheric Ozone https://sites.gsu.edu/geog1112/lab-4-2/
- Lab 3 The Troposphere https://sites.gsu.edu/geog1112/the-troposphere/
- NASA A Year in the Life of Earth’s CO2 (3:10 minutes) https://www.youtube.com/watch?v=x1SgmFa0r04
- An overview of the Earth's atmosphere http://www.groundinstructor.com/mod/page/view.php?id=970
2.2 Why Are Some Gases Called Greenhouse Gases?
The purpose of this section is to explain which atmospheric gases are considered greenhouse gases and why.
Why Are Some Gases Called Greenhouse Gases?
What are greenhouse gases?
Earth’s atmosphere is composed of a variety of gases. Some are greenhouse gases (GHGs) and others are not.
GHGs in Earth’s atmosphere include:
- Water
- Carbon Dioxide
- Methane
- Nitrous Oxide
- Ozone
- CFCs/HCFCs
- Sulfur Hexafluoride
While many people know that carbon dioxide and methane are important GHGs, it is less common for individuals to know that water is the primary GHG in our atmosphere. It keeps the earth’s temperature relatively constant, especially compared to other planets that have extreme cold and extreme hot temperatures.
Other gases in Earth’s atmosphere that are NOT GHGs include:
- Nitrogen
- Oxygen
- Argon
- Helium
- Neon
- Hydrogen
- Carbon Monoxide
These non greenhouse gases, plus water, make up over 99% of the gases in the atmosphere. Only tiny amounts of helium, neon, hydrogen, and carbon monoxide gases actually exist in the atmosphere.
Why are these NOT greenhouse gases?
The BIG question here is “Why are nitrogen, oxygen, argon, helium, neon, hydrogen, and carbon monoxide NOT GHGs?” Most people don’t know that there is a simple answer, and that answer is related to the symmetry of the molecules. In the images below the line of symmetry is shown with a green line.
Why Symmetry?
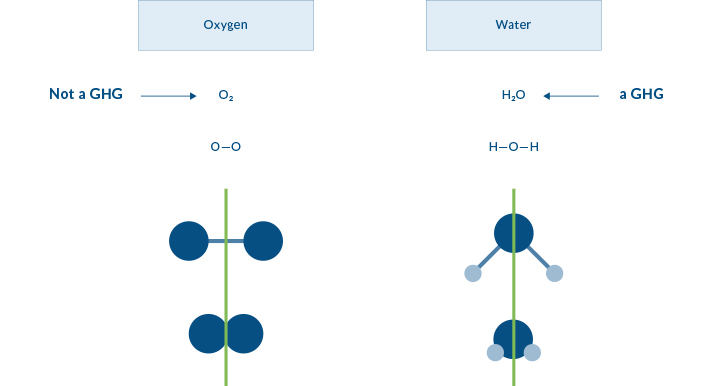
Atoms that make up the oxygen molecule (O2) include two oxygen atoms (O-O) joined together. In this illustration the ATOMS are represented by the balls, and the BOND joining the atoms are represented by sticks. Water (H2O) has two smaller atoms of hydrogen (shown as the lighter balls) joined to the oxygen atom in the middle by two bonds. Both oxygen and water form a symmetrical molecule, but one is a greenhouse gas and one is not.
Carbon Dioxide, CO2
Symmetrical or Unsymmetrical?
Carbon dioxide, whose structure is shown by these spheres, is also symmetrical.
So if these two GHGs are both symmetrical, how do they differ from non-GHGs? What about the symmetry is different?
The answer comes back to LIGHT!
Changes in the distance between atoms result when IR light is absorbed.
When longer lightwaves of infrared light (IR), shown as a yellow triangle in this illustration, hits a molecule’s bond, the bond stretches. A bond can be thought of as a spring between two balls. The IR light provides the energy that makes the bond stretch just as someone would have to pull on a spring to make it stretch. Stretching takes energy – from light.
CO2: Can its symmetry change?

So what happens to a carbon dioxide bond when IR light hits it? Just like other bonds, that bond stretches. If one bond stretches but not the other, the carbon dioxide molecule is no longer symmetrical. One bond is longer than the other so it loses its symmetry.
Only if symmetry changes can IR light be captured by the atmosphere
HERE IS THE BOTTOM LINE: A molecule is only a greenhouse gas if stretching can change its symmetry.
This happens because IR light can only change molecules that can absorb IR light energy. As it turns out, molecules whose symmetry does not change cannot absorb light in the IR region.
Does the symmetry of other gases change?
Hydrogen, Oxygen and Nitrogen are NOT greenhouse gases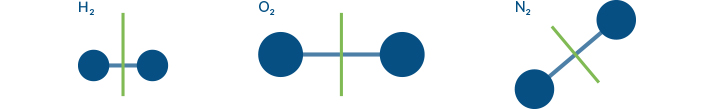
This illustration shows that the symmetry of these molecules cannot change. Even if the bonds were to stretch, they would not lose symmetry, so they cannot be GHGs.
These are NOT greenhouse gases
Of course molecules without bonds cannot change their symmetry at all, so none of these are GHGs.
Can water's symmetry change?
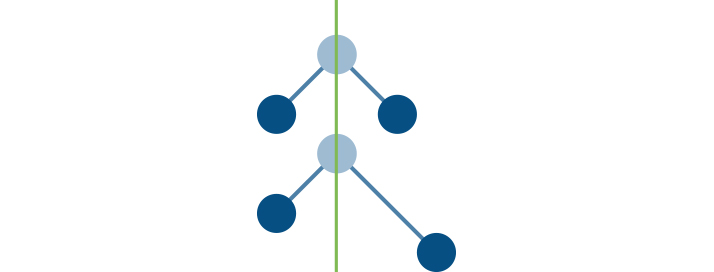
Water molecules can definitely change their symmetry so it is also an important GHG. However, there is naturally so much water in the atmosphere that the addition of more does not influence climate change quite like increases in carbon dioxide or other greenhouse gases.
Only molecules whose symmetry changes can be greenhouse gases
The key point to remember is this → only gases with molecules whose symmetry changes can be classified as greenhouse gases.
Why Are Some Gases Called Greenhouse Gases?
Why Are Some Gases Called Greenhouse Gases?
- Pacific Institute for Climate Solutions CO2 and the Greenhouse Effect (8:06 minutes) https://www.youtube.com/watch?v=sJ0eN_93l4k&feature=youtu.be
- Pacific Institute for Climate Solutions More than just CO2 (5:23 minutes) https://www.youtube.com/watch?v=fA9NwUiU4sU&feature=youtu.be
- Changing Climates, Colorado State University, Why Greenhouse Gases Make the Planet Warmer https://www.youtube.com/watch?v=AIBk0pGV_BQ
- An overview of GHGs: https://en.wikipedia.org/wiki/Greenhouse_gas
2.3 Global Warming Potential
The purpose of this section is to provide information about the characteristics of greenhouse gases that contribute to their capacity to impact global warming.
Global Warming Potential
Why are some greenhouse gases more potent than others?
The concentration level of a greenhouse gas in the atmosphere and the Global Warming Potential (GWP) of that gas combine to determine the impact it has on global warming.
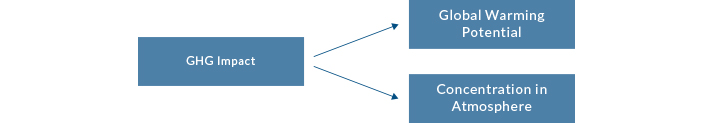
Carbon dioxide is the greenhouse gas most commonly discussed because its atmospheric concentration is increasing and the increase is largely attributed to human activity. Thus we can change behaviors to impact the concentration in the atmosphere. However, CO2 has a long life in the atmosphere, so urgency of action to reduce CO2 emissions is critical. An understanding of all greenhouse gases is necessary to appreciate the complex issue of the greenhouse effect and climate change.
Please view this video as an introduction to the topic:
Methane (CH4), an off-gas produced by decaying organic matter such as animal excrement and debris in landfills, is actually a more potent GHG than carbon dioxide. However, there is much less methane than carbon dioxide in the atmosphere and it resides in the atmosphere for a much shorter time than CO2. [On a related note: Scientists are concerned about increasing levels of atmospheric CH4 being influenced by a positive feedback mechanism as global warming is causing the release of methane trapped in arctic permafrost as it thaws due to warmer temperatures.]
The graphic below shows that there are two factors that contribut to the impact greenhouse gases have on global warming.
Global Warming Potential
Global Warming Potential (GWP) has the rather confusing definition of the “amount of warming that a gas will cause in the next 100 years, compared to the same volume of carbon dioxide."

This graphic uses methane (CH4) to illustrate how Global Warming Potential is conveyed.
In 100 years, a pound of methane (single circle) will have the same global warming effect as approximately 21 pounds of carbon dioxide (21 circles). This puts the Global Warming Potential of methane at 21 in this comparison to carbon dioxide. [The range of GWP values typically referenced for methane over 100 years is between 21 and 38.] For more information about Global Warming Potential see this website https://www.epa.gov/ghgemissions/understanding-global-warming-potentials
What contributes to Global Warming Potential?
Global Warming Potential for each gas in our atmosphere depends on two things:
- how much infrared (IR) light the gas absorbs and
- how long the gas stays in the atmosphere.
1) Absorbing IR
When infrared (IR) light strikes a bond, that bond absorbs the light and causes the bond to stretch. Some bonds can absorb more energy from the IR light than others. Compare this to what you know about the water absorbancy of cotton fibers versus polyester fibers. Cotton fibers naturally absorb more water than polyester fibers just like some molecular bonds can absorb more IR light than others.
2) How long gases remain in the atmosphere
Some global warming gases are more stable in the environment than others. A good analogy would be different degradation rates of paper and plastic when exposed to the outdoor environment. We expect both paper and plastic to deteriorate with environmental exposure, however we generally expect that paper will degrade faster than plastic when left outdoors. Similarly, some gases degrade faster than others. Global warming gas molecules in the atmosphere degrade because sunlight and other chemicals in the air break their bonds, or they go out into space, or they return to the surface of the earth. How long a gas stays in the atmosphere is called its residence time.
Global Warming Potential
The table below shows common greenhouse gases, their GWP, residence time in years, relative ability to absorb IR and common sources. The sources of GHGs related to textile production have been highlighted here.
| Gas | Global Warming Potential | Residence TIme (year) | Relative Ability to Absorb Infrared | Common Source |
|---|---|---|---|---|
| Carbon Dioxide CO2 |
1 | 50-200 | 1 | Fossil fuel burning |
| Methane CH4 |
21 | 12 | 43 | Agriculture, refining |
| Nitrous Oxide N4O |
310 | 120 | 250 | Agriculture, nylon |
| Chlorofluoro- carbons CFCs |
7,100 | 195 | 19,000 | Refrigerants |
| Perfluoro- carbons PFCs |
~9,000 | 2,600-4,000 | 21,000 | Stain resistant fabric finishes |
Notice the importance of these gases to the fabric and textile industries.Besides PFCs and N2O, the manufacture of all textiles requires energy to produce electricity and heat. Nearly all of the energy used in the industry is from fossil fuels. Methane is released in petroleum refining and fracking operations. In addition to energy, petroleum provides the raw materials for production of many textiles.
The residence time of several greenhouse gases are shown in the third column of this table. All of the years that these gases are in the atmosphere they are acting as global warming gases, absorbing IR light and contributing to increased temperatures.
Notice that carbon dioxide will stay in the atmosphere much longer than methane, and that PFCs, which are used on stain resistant fabrics, have very, very long residence times in the atmosphere. N2O is a compound that comes from the manufacture of nylon, and is important in agricultural based textiles and dyes. It also stays in the atmosphere a relatively long time of 120 years.
Finally, this table above shows the Global Warming Potential (GWP) of the GHGs listed. Remember that GWP is the amount of warming that a gas will cause in the next 100 years, compared to the same volume of carbon dioxide.It is important to point out that the global warming potential is a combination of the years the gas will reside in the atmosphere and the ability of the gas to absorb infrared light energy. Note for example, that the residence time of methane, CH4, is shorter than that for carbon dioxide, but it has a higher GWP because it strongly absorbs IR radiation.
Please view this video as a review of the content related to Global Warming Potential.
Global Warming Potential
Global Warming Potential
- Pacific Institute for Climate Solutions Examples of Global Warming (7:09 minutes) https://www.youtube.com/watch?v=VttL3ZYQpy4
- EPA Climate Change website https://www.epa.gov/climate-change
2.4 Carbon Cycle
The purpose of this section is to provide an understanding of the carbon cycle and how changes in carbon stores impact climate change.
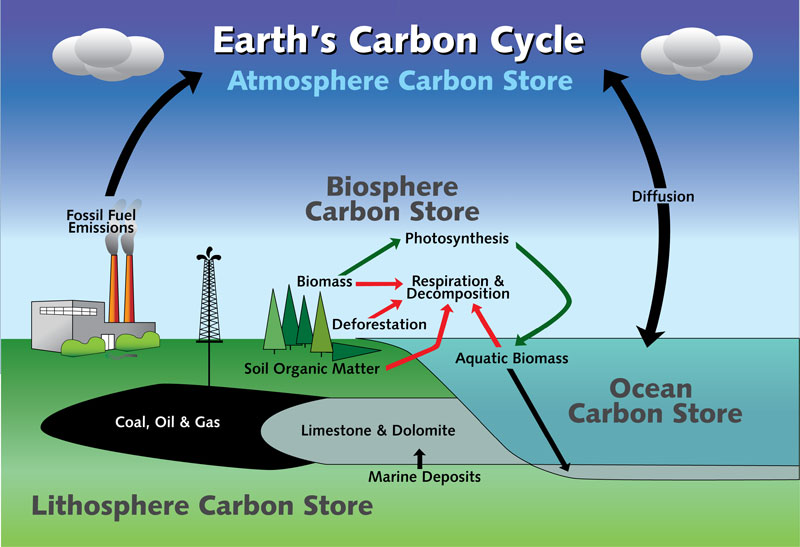
Carbon Cycle
Source: http://www.dec.ny.gov/energy/76572.html
The earth has five spheres that are part of the carbon cycle:
- Atmosphere (envelope of gases surrounding earth)
- Hydrosphere (all bodies of water)
- Cryosphere (frozen part of earth’s surface)
- Lithosphere (rigid outer part of earth’s surface)
- Biosphere (regions of earth’s surface occupied by living organisms)
All living organisms are built of carbon compounds.It is the fundamental building block of life and an important component of many chemical processes.
In the atmosphere, carbon is present primarily as carbon dioxide (CO2), but also as other less abundant but climatically significant gases, such as methane (CH4).
Carbon is exchanged, or "cycled" among Earth's oceans, atmosphere, ecosystem, and geosphere.This movement of carbon, in its many forms, is the carbon cycle.
The video below is a simple explanation of the carbon cycle that students might enjoy.
Under In the Classroom below there is a classroom activity the provides a way for students to discover more about the carbon cycle.
These are key points for students to know about the carbon cycle:
- Life is fueled by carbon compounds.
- Animals and plants give off (release) carbon dioxide during respiration.
- Plants use carbon dioxide during photosynthesis and build new carbon compounds.
- CO2 is released by the burning of fossil fuels.
- The atmosphere and oceans are continuously exchanging CO2.
- The carbon cycle consists of reservoirs that store carbon. The storage reservoirs include the atmosphere, the oceans, vegetation, rocks, and soil.
- The carbon cycle also consists of the processes by which carbon moves between these reservoirs.
- If more carbon enters a pool than leaves it, that pool is considered a carbon sink. If more carbon leaves than enters, it is a carbon source.
- Carbon dioxide is a major player in the Earth’s energy balance. See 5.1 Climate System for more about the Earth's energy balance.
Carbon Cycle
About POGIL Activities
You can learn more about POGIL at: https://pogil.org/about
Carbon Cycle
- The Department of Geosciences at Georgia State University has published a set of labs that may be helpful. Of particular interest are the following:
- Lab 4 The Carbon Cycle (Part 1) https://sites.gsu.edu/geog1112/lab-4-2/
- Lab 4 The Carbon Cycle (Part 2) https://sites.gsu.edu/geog1112/lab-4-the-carbon-cycle-part-2/
- TED Talk The Carbon Cycle (3:54 minutes) https://ed.ted.com/lessons/the-carbon-cycle-nathaniel-manning by Nathaniel Manning and Jill Johnston
3. Anthropogenic Contributors to Climate Change
This section describes some of the most common anthropogenic (human) sources of greenhouse gases.
Anthropogenic Contributors to Climate Change
Since the start of the Industrial Revolution, humans have engaged in a range of behaviors (e.g., burning fossil fuels as an energy source and deforestation) that have increased the release of carbon dioxide into the atmosphere. Scientific consensus is that these behaviors are interrupting the carbon cycle and contributing to long-term increases in the amount of greenhouse gases in the atmosphere.
We are releasing carbon from ancient carbon stores so quickly that the natural processes in place to capture and store it cannot keep up with the release. As a result, there is an imbalance in carbon dioxide levels in the Earth's spheres. This imbalance of CO2 levels is mostly in the atmosphere and hydrosphere.
Additionally, as we cut down large sections of forest we are removing one of the major processes in the carbon cycle designed to remove CO2 from the atmosphere (through the photosynthesis of the trees).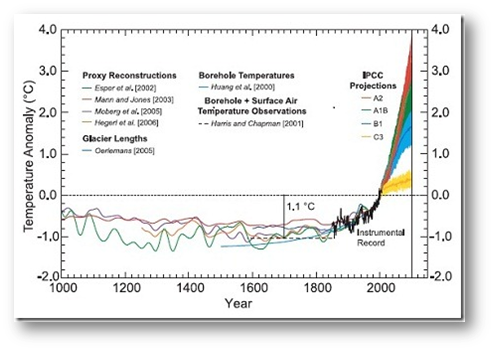
Source: sedgemore.com/2010/10/consolidating-the-climate-change-record/
The graphic above is popularly known as the “hockey stick” graph of global temperature change over the past 1000 years. The figure clearly illustrates the drastic nature of recent climate change and the rapid change in mean global temperatures since the mid-1800s.
Recent Climate Trends
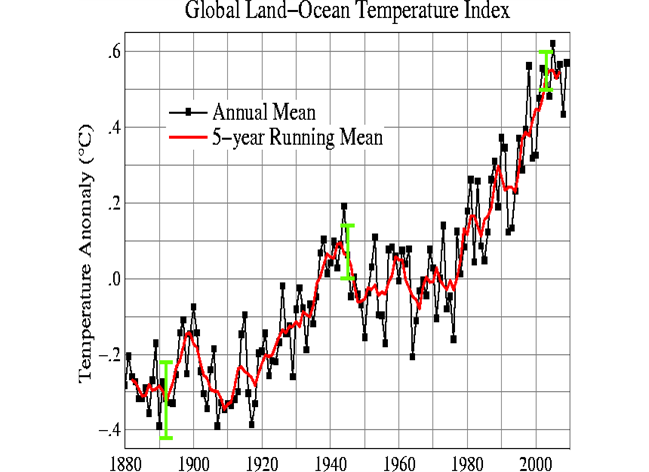
Source: http://data.giss.nasa.gov/gistemp/graphs_v3/
This graphic shows the annual mean global temperatures since 1880.
There are many different factors – some natural and some not – contributing towards global climate change. However, the scientific community widely agrees that, "Human influence on the climate system is clear, and recent anthropogenic emissions of greenhouse gases are the highest in history. Recent climate changes have had widespread impacts on human and natural systems."
Source: IPCC, 2014: Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change [Core Writing Team, R.K. Pachauri and L.A. Meyer (eds.)]. IPCC, Geneva, Switzerland, p 2
Scientific consensus is clear that by altering the composition of the atmosphere humans are increasing the average temperature of the Earth’s surface.
Recent Greenhouse Gas Trends
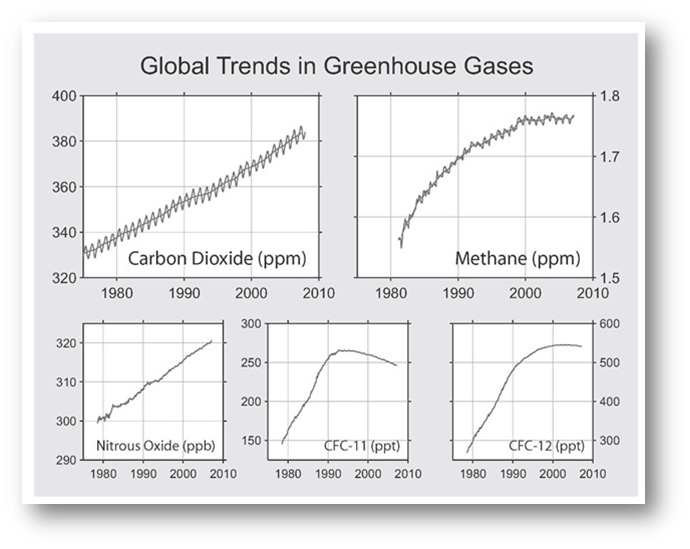
Source: http://www.globalwarmingart.com/wiki/File:Major_greenhouse_gas_trends_png
The rise of GHGs are believed to be responsible for most of the increase in global average temperatures during the last 50 years.
At present, approximately 99 percent of the 100-year global warming potential for all new emissions can be ascribed to just these three gases: carbon dioxide, methane, and nitrous oxide.
Intergovernmental Panel on Climate Change
According to the IPCC Fourth Assessment Report: Climate Change 2007:
“Warming of the climate system is unequivocal, as is now evident from observations of increases in global average air and ocean temperatures, widespread melting of snow and ice, and rising global average sea level.” https://www.ipcc.ch/publications_and_data/ar4/syr/en/spms1.html
“Most of the observed increase in global average temperatures since the mid-20th century is very likely* due to the observed increase in anthropogenic greenhouse gas concentrations.” https://www.ipcc.ch/publications_and_data/ar4/wg1/en/spmsspm-understanding-and.html#footnote12
*IPCC defines ‘very likely’ as greater than 90 percent probability of occurrence.
Ninety-seven percent of climate scientists agree that climate-warming trends over the past century are very likely due to human activities, and most of the leading scientific organizations worldwide have issued public statements endorsing this position.
So what are humans doing to induce climate change?
The combination of exploding population, economic growth and 150 years of industry based on fossil fuels has brought about our rapidly warming climate.
A massive amount of carbon has been sequestered or stored underground, in many forms including coal, oil, and natural gas, which we are now burning for transportation, power generation, and industrial process.
Millions of years ago decaying organic matter was buried by sediments and in the heat and pressure underground became coal, oil, and natural gas.
When these materials are burned for energy, it releases the sequestered carbon and throws Earth’s carbon cycle out of balance.
Electricity Generation
Many students are unaware of the relationship between electricity generation and carbon dioxide production. This section provides an overview.
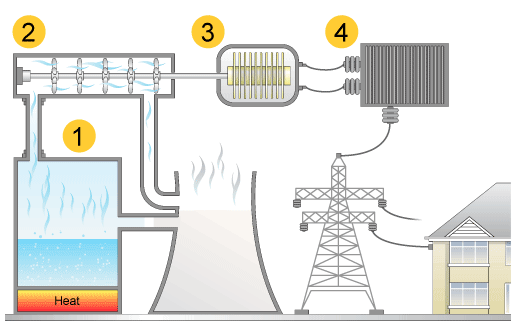
Source: http://www.bbc.co.uk/schools/gcsebitesize/science/21c/sustainable_energy/generating_electricityrev4.shtml
Electrical generation and distribution of electricity to consumers most commonly includes the components in this diagram.
- Steam Generation (water boiled using coal or other sources of energy)
- Turbine (steam causes the turbine to spin which translates into mechanical, or kinetic, energy transferred to the generator)
- Electromechanical Generator (transforms kinetic energy into electricity)
- Transformer (reduces electricity wattage for use by consumers)
Electrical Energy Sources
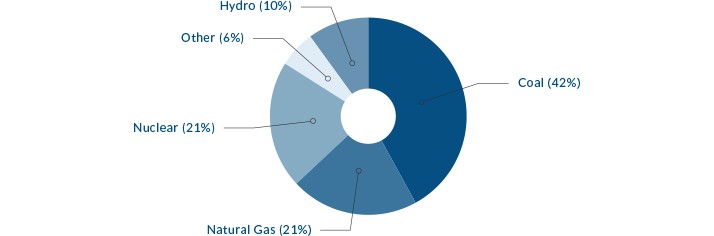
There are many different ways to heat water to create steam to produce electricity. The chart above show the sources of energy used in the United States to produce electricity. Coal is the most common energy source used to generate electricity in the United States.
Globally, more than 40% of all carbon dioxide emissions come from burning coal.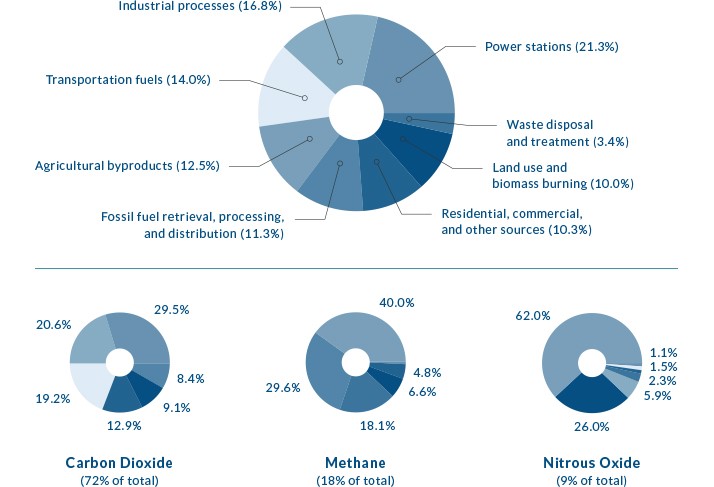
Source: http://www.globalwarmingart.com/wiki/File:Greenhouse_Gas_by_Sector_png
Anthropogenic Contributors to Climate Change
Anthropogenic Contributors to Climate Change
- This link is to a report by the International Energy Agency on Energy and Climate Change https://www.iea.org/publications/freepublications/publication/WEO2015SpecialReportonEnergyandClimateChange.pdf
- Pacific Institute for Climate Solutions Human Influence (4:08 minutes) https://www.youtube.com/watch?v=-GDY3dCqWQU
- Pacific Institute for Climate Solutions What YOU Can Do About Climate Change (8:59 minutes) https://www.youtube.com/watch?v=VTfgNFz1DBM
- 100 Views of Climate Change, Colorado State University http://changingclimates.colostate.edu/
- Science Clarified Chapter 1 The Development of Energy http://www.scienceclarified.com/scitech/Energy-Alternatives/The-Development-of-Energy.html
- An overview of energy storage and transfer with information about energy sources http://www.bbc.co.uk/bitesize/ks3/science/energy_electricity_forces/energy_transfer_storage/revision/8/
- A slide show on energy generation from multiple sources https://www.slideshare.net/slideshow/energy-resources-types-35381452/35381452